Isolation of Rabbit Cardiomyocytes
Stephen C Armstrong, Ph.D., Asst Professor,
Dept of Pathology,
East Tenn. State Univ,
Johnson City TN ,
Email: armstron@access.etsu.edu
Olaf Oldenburg, M.D.
Dept of Physiology
University of South Alabama
Mobile AL
Email: ooldenburg@usamail.usouthal
Introduction
We have experience isolating rat, rabbit and pig
cardiomyocytes. Each of these animal models have their distinct
advantages and disadvantages and these must be taken into account
when choosing the species for your model. The factors to consider
include: Labor intensivity of the isolation procedure, cost
efficiency, cell yield and relevance to the human model. From my
experience, rabbit cardiomyocytes are a good compromise and
balance of these logistical and scientific considerations. Rats
are cheaper and pigs give a high cell yield and are potentially
more relevant to the human model. However, the isolation of
rabbit cardiomyocytes is relatively inexpensive and is not as
labor intensive as the isolation of pig cardiomyocytes,
delivering a higher cell yield than the rat model. The rabbit
model does not share the distinction of the rat model, in which
adenosine does not mimic cardioprotection. The rabbit is
intermediate between the rat and pig in terms of heart rate and
action potential duration and by these criteria the pig is more
closely related to the human. We have found distinctions between
the rabbit and pig cardiomyocytes in terms of their responses to
our in vitro models of ischemia and the delay of osmotic
fragility by ischemic preconditioning(IPC). The onset of osmotic
fragility is defined as the sarcolemmal rupture and inability to
exclude trypan blue after a brief resuspension of ischemic cells
in a hypotonic (85mOsm) buffer. Osmotic fragility has been
proposed to be a correlate of irreversible ischemic injury1-3.
After 60 min. of ischemia, rabbit cardiomyocytes contract
concurrent with ATP depletion and osmotic fragility occurs 30-60
min. after the onset of cell contracture. The onset of osmotic
fragility in pig cardiomyocytes occurs concurrent with cell
contracture 4. In contrast to rabbits, in which in vitro IPC does
not alter cell contracture, subjection of pig cardiomyocytes to a
IPC protocol, protects cells by delaying cell contracture and
thus the onset of osmotic fragility. I will present the details
of our cell isolation methods and our in vitro models of ischemia
and ischemic preconditioning.
Cell Isolation
Isolated, calcium tolerant, adult rabbit cardiomyocytes are
prepared by collagenase perfusion as previously described 5according
to the methods of Hohl et. al. 6, with the specific
modification that adenosine is excluded from all media. Our
calcium-free perfusion buffer contains in mM, NaCl (125), MgSO4
(1.18), KCl (4.75), KH2PO4 (1.2 ), HEPES (30)
bovine serum albumin (BSA, fraction V) (1g/liter), glucose (11),
taurine (58.5) creatine (24.9) EGTA (0.02)*, plus a complete
amino acid mixture (MEM
Amino Acid Solution (Gibco, 20ml/L of 50x stock) and MEM
Non-Essential Amino Acid Solution (Gibco,10ml/L of 100x
stock) and vitamin
solution (Sigma, 10ml/L of 100x stock). The mixture is
bubbled with a 95% O2 / 5% CO2 mixture for
30 min and then the pH is adjusted to 7.2 before filtering. This
perfusion buffer is then filtered through a 5 µm cellulose
acetate filter. You will need about a liter for each
isolation.
Our anesthesia consists of 50 mg/ml pentobarbital I.V. in
animal that have been heparinzed with 1000U/Kg heparin I.P., 10
min prior to anesthesia. The thoracic cavity is entered and hearts
are excised and placed in 200 ml of ice cold perfusion buffer and
the aorta is quickly isolated and mounted on a Langendorff
apparatus. The heart is flushed for 5 min in a non-recirculating
mode with 37º C oxygenated (95% O2/5% CO2)
perfusion buffer. The heart is perfused at 80 - 100 cm H20
by gravity.
After a 5 min equilibration period perfusion is switched to a
recirculating collagenase perfusion using a peristaltic pump. The
perfusate consists of 200 ml of the above described perfusion
buffer to which 200 U/ml collagenase (Worthington, Type II)
is added and then filtered through a 5 µm cellulose acetate
filter. We use Worthington Type II collagenase and several lots
are tested periodically to determine which lot will give the
ideal preparation of cells (samples free of charge are available
from Worthington Inc.). Even if these enzymes are called "collagenase"
they consists of a mixture of collagenase and caseinase, and have
clostripain and tryptic activities. In general I prefer
collagenase lots that have relatively low activities of
collagenase (200-250 U/mg). After switching to the collagenase
perfusion, pH (7.3 – 7.4) and coronary pressure are
continuously monitored. Adjust as needed with NaOH. After the
collagenase reacts with the heart for 3-5 min there is a fall in
the coronary pressure and the flow rate should be increased to
restore the pressure to 80 -100 cmH2O. Perfusion is
continued for 15-20 minutes until the heart starts to fall apart.
Ventricles are then removed and minced in 50ml of collagenase
perfusate containing to which BSA has been added to achieve a
final concentration of 2%. Disperse the cells by flushing
them in and out of a large bore pipette (e. g. a plastic transfer
pipette from which the tip has been cut off) 10 to 15 times.
Cells are incubated in this mincing buffer for 10 min using a
shaker bath at 37° C and bubbled with 95% O2 / 5%
CO2. The next step is a nylon mesh filtration (200
– 350m M pore diameter). Cells are pelleted by a 1.5 min 200
rpm centrifugation (for this use four 15ml tubes with 12.5ml cell
suspension in each) and then resuspended in 20 ml of wash buffer,
containing in mM, NaCl (125), KCl (4.75), KH2PO4
(1.2), MgSO4 (1.2), HEPES (30), glucose (11), and 2%
BSA supplemented with creatine, taurine, vitamins and amino acids
as above but without EGTA or Ca2+. You need about 100ml
of this buffer for the purification steps. Filter the wash buffer
through a 5 µm cellulose acetate filter. Cells are incubated in
this wash buffer for 30 minutes in a 100 ml Erlenmeyer flask in a
37°C shaker water bath at 25 shakes/min to allow re-establishment
of normal electrolyte gradients. Supply oxygen by gently blowing
95% O2 / 5% CO2 over the surface. The 30
min incubation is followed by addition of five aliquots of 100ul
of 50 mM CaCl stock solution at 5 min intervals to achieve a
final concentration of 1.25 mM.
For purification of viable calcium tolerant cells, divide the
cell suspension equally into two 15ml tubes. Leave those tubes on
the bench top for approximately 5min to get an initial
sedimentation pellet, discard the supernatant. This step is
followed by two brief centrifugations of approximately 200 rpm, 1
– 1.5 min each. After each spin discard the supernatant and
resuspend the pellet into fresh calcium-containing wash buffer (add
calcium to the above wash buffer to achieve 1.25mM Ca2+;
pH 7.4), gently mix the cells with the fresh buffer before each
centrifugation. Isolates averaged 45 million cells, 75-80
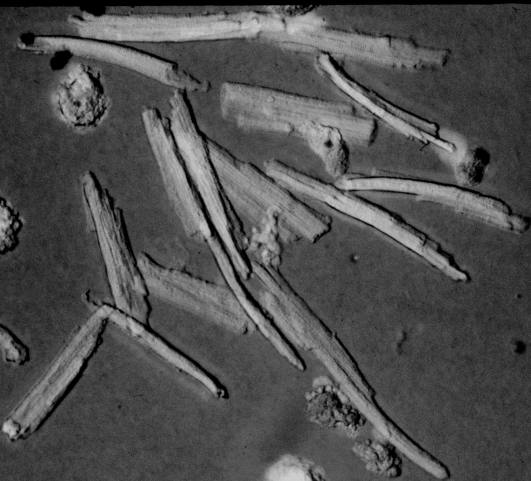
percent of which are rod-shaped (see figure 1).

Figure 1
Oxygenated
cardiomyocytes are elongated with prominent striations indicating
the relaxed state of sarcomeres . These cells exclude trypan blue
and do not have blebs.
Ischemia model and assessment of cell injury:
An aliquot of the cell suspension is placed in a 1.8 ml
microcentrifuge tube (Fisher Scientific, Pittsburgh, PA) and
centrifuged into a pellet. Each cell pellet occupies a volume of
about 0.25 ml, and measures 0.8-1 cm in thickness. Excess
supernatant is removed to leave a fluid layer above the pelleted
cells of about one third the volume of the pellet. After layering
with mineral oil, the cell pellets were incubated without
agitation at 37°C. A 25 µl sample of the final cell pellet is
removed through the oil layer, with care to prevent introduction
of air, at the appropriate time points and resuspended for 3-5
minutes in 200 µl of hypotonic (85 mOsm) buffer containing 3 mM
amytal as a mitochondrial inhibitor, to preclude cell rounding
due to reoxygenation during cell swelling. A 25 µl sample is
mixed with an equal volume of counting media (0.5% glutaraldehyde
in 85 mOsM modified Tyrodes solution, with reduced NaCl,
containing 1% trypan blue). Microscopic examination at 100X
magnification determined the morphology (rod, round or square (See
Figure 2) and the permeability of the cells to trypan blue
(See Figure 3) as described previously7, 8.

Figure 2
Ischemic contracture
of rabbit cardiomyocytes occurs after 60 minutes of
ischemia, concurrent with ATP depletion. Dome shaped, sub-sarcolemmal
blebs are observed at this point of ischemia following
resuspension in hypotonic buffer.
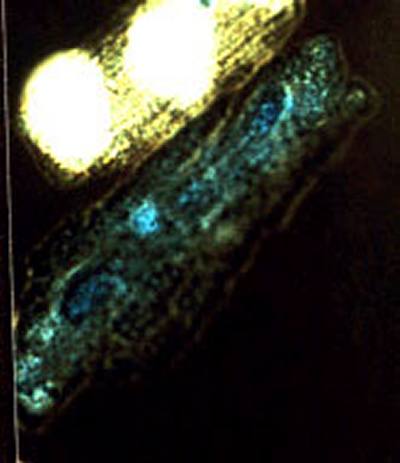
Figure 3
At 120 minutes of
ischemia, osmotic fragility of cardiomyocytes is observed by the
inability of cells to exclude trypan blue after resuspension in a
hypotonic buffer.
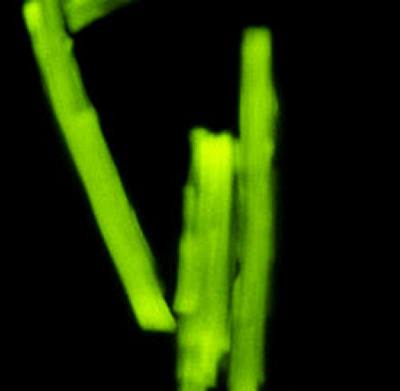
Figure 4-
High levels of ATP in
oxygenated rabbit cardiomyocytes can be indirectly assessed by
the mitochondrial potential fluorescent indicator, Rhodamine 123.
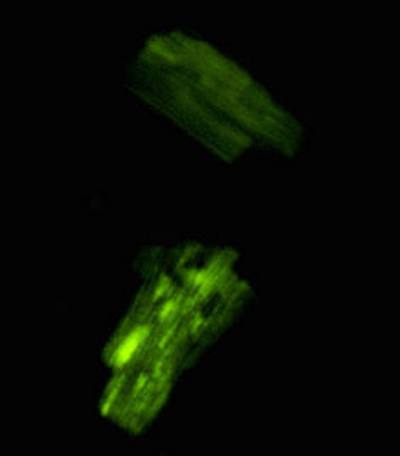
Figure 5-
ATP depletion is
observed in the contracted (square shaped) , ischemic cells.
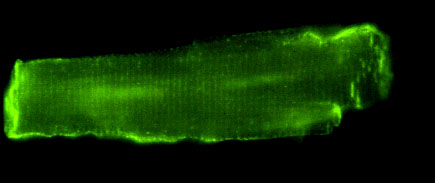
Figure 6
Immuno-staining of
oxygenated cardiomyocytes for vinculin shows a costameric (rib-like)
pattern at the sarcolemma and staining at the intercalated disks.
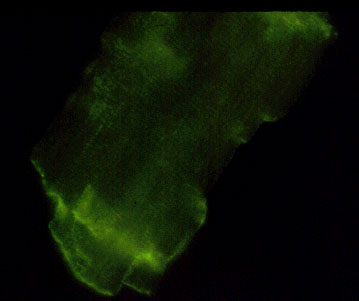
Figure 7
Immuno-staining of
ischemic cardiomyocytes for vinculin shows that the level of
fluorescence at the sarcolemma and intercalated disk is
diminished 9.
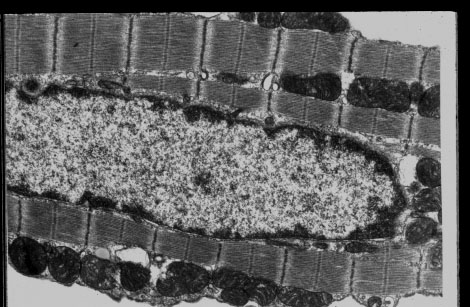
Figure 8
Transmission electron microscopy of oxygenated rabbit
cardiomyocytes demonstrates the relaxed state of the sarcomeres
and dense, unswollen mitochondria, similar to oxygenated
myocardium. The nuclear chromatin is dispersed and the sarcolemma
does not show blebs.
References
1. Steenbergen CJ, Hill ML, Jennings RB. Volume regulation and
plasma membrane injury in aerobic, anaerobic and ischemic
myocardium in vitro : Effect of osmotic swelling on plasma
membrane integrity. Circ Res 1985;57:864-875.
2. Tranum-Jensen J, Janse MJ, Fiolet JWT, Krieger JG,
D'Alnoncourt CN, Durrer D. Tissue osmolality, cell swelling, and
reperfusion in acute regional myocardial ischemia in the isolated
porcine heart. Circ Res 1981;49:364-381.
3. Ganote CE, Vander Heide RS. Irreversible injury of isolated
adult rat myocytes: Osmotic fragility during metabolic inhibition.
Am J Path 1988;132:212-222.
4. Armstrong SC, Kao R, Gao W, et al. Comparison of in
vitro preconditioning responses of isolated pig and rabbit
cardiomyocytes: effects of a protein phosphatase inhibitor,
fostriecin. J Mol Cell Cardiol 1997;29:3009-3024.
5. Vander Heide RS, Angelo JP, Altschuld RA, Ganote CE. Energy
dependence of contraction band formation in perfused hearts and
isolated adult myocytes. Am J Path 1986;125:55-68.
6. Hohl CM, Altschuld RA, Brierley GP. Effects of calcium on
the permeability of isolated adult rat cells to sodium and
potassium. Arch Biochem Biophys 1982;221:197-205.
7. Armstrong SC, Ganote CE. Effects of 2,3-butanedione
monoxime (BDM) on contracture and injury of isolated rat myocytes
following metabolic inhibition and ischemia. J Mol Cell
Cardiol 1991;23:1001-1014.
8. Vander Heide RS , Rim D, Hohl CM, Ganote CE. An in vitro
model of myocardial ischemia utilizing isolated adult rat
myocytes. J. Mol Cell Cardiol 1990; 22:165-181
9. Armstrong, SC and Ganote CE. Flow cytometric analysis of
isolated adult cardiomyocytes: Vinculin and tubulin fluorescence
during metabolic inhibition and ischemia. J Mol Cell Cardiol
1992; 24: 149-162